中文名称:FITC标记链霉亲和素
英文名称:FITC-conjugated Streptavidin
其他别称:FITC-conjugated Streptavidin
FITC标记Streptavidi链霉亲和素(1.0 mg)
Streptavidin-FITC
Streptavidin-fluorescein
外观:淡黄色溶液
分子量:60kda
规格:ml
纯度:≥95%
浓度:0.5mg/ml
质量控制:95%
Abs/Em最大值:488/520nm
储存条件:4℃
保存时间:1 year
用途:荧光标记等
产地:上海
链霉亲和素与生物素具有很高的亲和力。异硫氰酸链霉亲和素(FITC)可用于检测生物素化抗体。用488nm激光激发FITC,其最大发光强度为520nm。
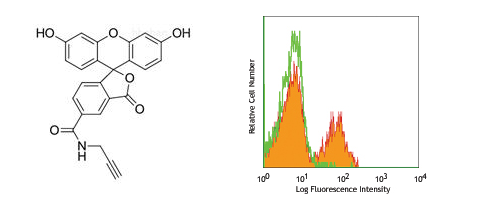
描述:
本制品以大肠杆菌表达,经生物素亲和纯化的基因重组链霉亲和素蛋白和FITC标记链霉亲和素 FITC-Streptavidin为原料,采用化学交联法标记制备。本产品溶于PBS(pH7.3)中,蛋白浓度≥1mg/ml,含有稳定剂,可用于生物素及生物素化标记物的检测与分析。本产品需经稀释方可使用,最佳工作浓度请自行优化。
链霉亲和素(Streptavidin,SA,SA V1,SA V2,Streptavidin V1,Streptavidin V2)别称链霉亲合素、链亲和素或链亲合素,是具有与卵亲和素(Avidin,AV)相似生物学特性的一种蛋白质,都能与4分子的生物素(Biotin)特异性结合,结合常数为1×10-15/M。同时,由于SA等电点低,不带糖基或糖链,在检测中具有比卵亲和素更低的背景,故在检测中的灵敏度和特异性都高于卵亲和素,建立在此基础上的生物素-链霉亲和素系统的应用比生物素-亲和素系统有更大优势。
相关产品:
Neutravidin-thiol
Streptavidin
Streptavidin-CY5.5
Streptavidin-CY5
Streptavidin-CY3
Streptavidin-TRITC
Streptavidin-thiol
Avidin-CY3
Avidin-CY5
Avidin-CY5.5
Avidin (without modification)
Avidin-Sepharose
Avidin-thiol
Avidin-FITC
购买须知:
1.关于颜色
产品因不同产品的分子量不同,产品性状和颜色会有差别。
2.关于客服
如您的咨询没能及时回复,可能是当时咨询量过大或是系统故障。
我们将提供售后服务。
3.关于发货
我们的合作快递公司有顺丰、圆通、申通、韵达。
产地:上海
纯度:99%
用途:仅用于科研